近日,求臻医学生信团队助力合作伙伴设计并实现了一款基于Docker的FLT3-ITD检测框架,该框架能够准确检测FLT3-ITD,检测结果与一代测序结果的一致性高达99%以上,突破了现有NGS方法低估突变频率的核心技术难题。目前,该研究成果已在国际生物信息学领域重要科技期刊Briefings in Bioinformatics(SCI影响因子:8.99,中科院JCR数学与计算生物学1区Top期刊)上在线发表。此项研究适用于急性髓系白血病(AML)患者,可有效指导AML患者预后分层和临床个性化诊疗。

研究背景
FLT3-ITD是急性髓系白血病的预后不良指标之一,伴有该突变的患者临床上常表现为预后差、复发率高且总生存期短。目前临床上广泛采用PCR和毛细管电泳法检测FLT3-ITD,并通过片段分析对其长度和突变频率进行定量评估。但由于该方法存在操作过程易污染,无法识别突变位置和碱基序列,以及突变峰重叠导致FLT3-ITD个数识别出现偏差等不足,从而无法准确定位和量化FLT3-ITD。
二代测序(NGS)技术的蓬勃发展推动了FLT3-ITD检测软件的研发和应用。基于NGS的检测方法能够检测FLT3-ITD的突变位置、碱基序列、插入长度和突变频率等定量信息,有效地克服了传统检测方法的局限性。然而,基于NGS的检测方法仍存在安装部署复杂、检测结果假阳性较高且低估突变频率等问题。因此,突破以上技术瓶颈,开发基于Docker的FLT3-ITD自动化检测框架具有巨大的临床应用价值。
研究设计
本研究纳入了AML队列中的163例患者,使用求臻医学ChosenHeme®对血液样本进行基因检测。此外,基于二代测序数据模拟软件wgsim生成共计500例FLT3-ITD阳性样本。研究首先对现有NGS检测方法的原理、功能和局限性进行了全面研究,然后分别在模拟数据和真实样本上,围绕定性和定量分析能力、结果可读性以及运行时间,对Pindel、ITDseek、ScanITD、ITDetector、getITD和FLT3_ITD_ext六款代表性软件进行性能评估。

图1. 文章结构框架图
研究结果
基于以上对比分析结果,研究者设计了一款基于Docker的FLT3-ITD检测框架,实现了FLT3-ITD的精准检测,并使检测流程趋于自动化、可定制化以及跨平台可移植,为临床医生和科研人员选择合适的FLT3-ITD检测软件提供了有效的指导,同时进一步助力AML的预后分层和个性化诊疗。
未来,求臻医学将使用自主研发的ChosenHeme®血液系统肿瘤基因检测Panel,持续对大规模AML样本集进行测序,积累大量高通量测序数据,深度挖掘AML患者队列中FLT3-ITD的突变特征。与此同时,还将进一步关注靶向FLT3-ITD药物的筛选和研发,推动靶向治疗的发展进程,为更多的AML患者带来福音。
关于FLT3-ITD
FMS样酪氨酸激酶3(FLT3)基因位于染色体13q12.2,其编码的FLT3蛋白属于III型受体酪氨酸激酶(RTK)家族,可以调节细胞凋亡、增殖及造血干/祖细胞的分化,在造血过程中发挥重要作用。

图2. FLT3蛋白结构
FLT3-ITD,即FLT3基因内部串联重复,常见于FLT3基因的14号和15号外显子,其插入长度为3到几百个碱基对不等,多数情况下以3的倍数出现。每个患者中携带FLT3-ITD突变的个数一般为1-3个,且插入位置多覆盖14号外显子的Y591-Y597区域。FLT3-ITD的突变模式,如突变个数、插入长度以及突变频率等,不仅是AML的预后指标,还是AML风险分层的重要考虑因素,对其治疗方案的选择具有一定指导意义。
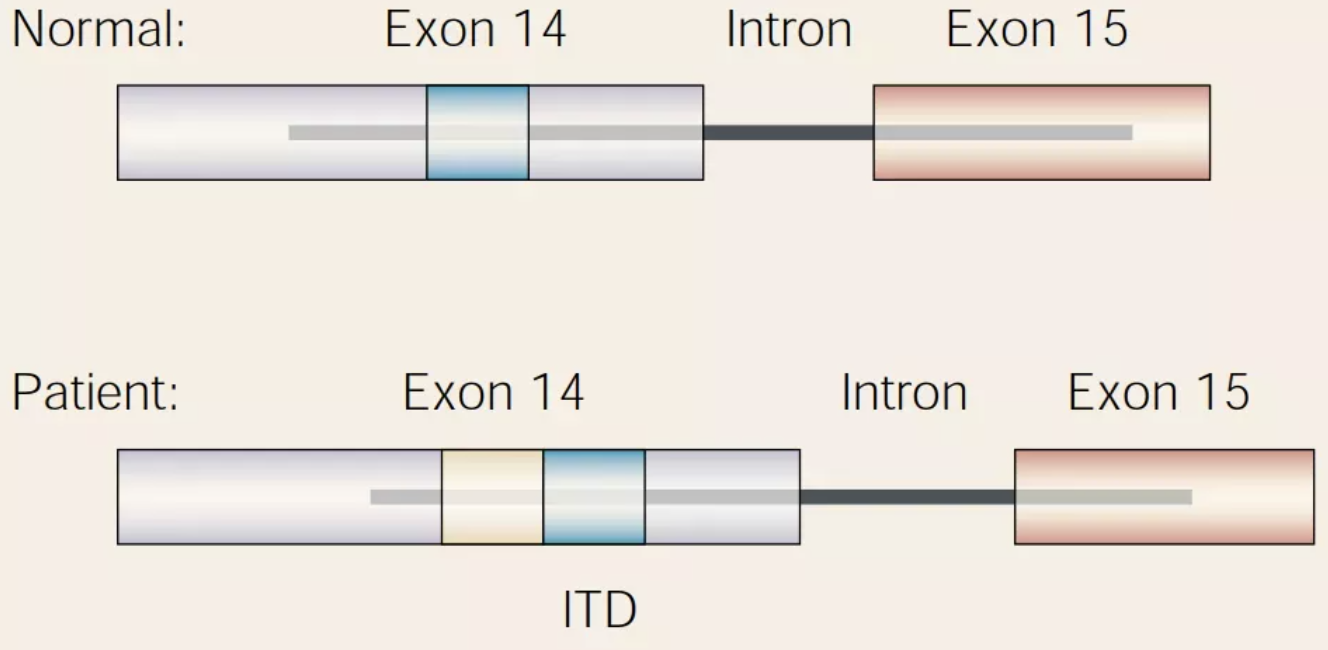
图3. FLT3-ITD突变特征
参考文献:
1. Lagunas-Rangel FA, Chávez-Valencia V. FLT3–ITD and its current role in acute myeloid leukaemia. Med Oncol 2017;34:114.
2. Siegel RL, Miller KD, Jemal A. Cancer statistics, 2016. CA Cancer J Clin 2016;66:7-30.
3. Papaemmanuil E, Gerstung M, Bullinger L et al. Genomic Classification and Prognosis in Acute Myeloid Leukemia. New Engl J Med 2016;374:2209-2221.
4. Döhner H, Estey E, Grimwade D et al. Diagnosis and management of AML in adults: 2017 ELN recommendations from an international expert panel. Blood 2017;129:424-447.
5. Daver N, Schlenk RF, Russell NH et al. Targeting FLT3 mutations in AML: review of current knowledge and evidence. Leukemia 2019;33:299-312.
6. Huang Y, Hu J, Lu T et al. Acute myeloid leukemia patient with FLT3-ITD and NPM1 double mutation should undergo allogeneic hematopoietic stem cell transplantation in CR1 for better prognosis. Cancer Manag Res 2019;11:4129-4142.
7. Fleischmann M, Schnetzke U, Schrenk KG et al. Outcome of FLT3-ITD-positive acute myeloid leukemia: impact of allogeneic stem cell transplantation and tyrosine kinase inhibitor treatment. J Cancer Res Clin Oncol 2017;143:337-345.
8. Kim Y, Lee GD, Park J et al. Quantitative fragment analysis of FLT3-ITD efficiently identifying poor prognostic group with high mutant allele burden or long ITD length. Blood Cancer J 2015;5:e336.
9. Lyu M, Liao H, Shuai X et al. The prognosis predictive value of FMS-like tyrosine kinase 3-internal tandem duplications mutant allelic ratio (FLT3-ITD MR) in patients with acute myeloid leukemia detected by GeneScan. Gene 2020;726:144195.
10. Liu S-B, Dong H-J, Bao X-B et al. Impact of FLT3-ITD length on prognosis of acute myeloid leukemia. Haematologica 2019;104:e9-e12.
11. Abou Dalle I, Ghorab A, Patel K et al. Impact of numerical variation, allele burden, mutation length and co-occurring mutations on the efficacy of tyrosine kinase inhibitors in newly diagnosed FLT3- mutant acute myeloid leukemia. Blood Cancer J 2020;10:48.
12. Schetelig J, Rollig C, Kayser S et al. Validation of the ELN 2017 Classification for AML with Intermediate Risk Cytogenetics with or without NPM1 -Mutations and High or Low Ratio FLT3-ITD s. Blood 2017;130:2694-2694.
13. Mekawy MA, Eissa DS, Hamza MT et al. FLT3 receptor/CD135 expression by flow cytometry in acute myeloid leukemia: Relation to FLT3 gene mutations and mRNA transcripts. Egypt. J. Medical Hum. Genet. 2018;19:345-351.
14. He R, Devine DJ, Tu ZJ et al. Hybridization capture-based next generation sequencing reliably detects FLT3 mutations and classifies FLT3-internal tandem duplication allelic ratio in acute myeloid leukemia: a comparative study to standard fragment analysis. Mod Pathol 2020;33:334-343.
15. Schumacher JA, Holgard VD, Sial F et al. Detection and Quantification of FLT3 Internal Tandem Duplication Mutations Do Not Vary Significantly Between Whole Blood and Blast-Enriched Samples. Am. J. Clin. Pathol. 2020;153:251-257.
16. Banasik M, Stanisławska-Sachadyn A, Sachadyn PJJoag. A simple modification of PCR thermal profile applied to evade persisting contamination. J. Appl. Genet. 2016;57:409-415.
17. Schranz K, Hubmann M, Harin E et al. Clonal heterogeneity of FLT3-ITD detected by high-throughput amplicon sequencing correlates with adverse prognosis in acute myeloid leukemia. Oncotarget 2018;9:30128-30145.
18. Kim B, Kim S, Lee S-T et al. FLT3 internal tandem duplication in patients with acute myeloid leukemia is readily detectable in a single next-generation sequencing assay using the pindel algorithm. Ann. Lab Med 2019;39:327-329.
19. Bibault J-E, Figeac M, Hélevaut N et al. Next-generation sequencing of FLT3 internal tandem duplications for minimal residual disease monitoring in acute myeloid leukemia. Oncotarget 2015;6:22812.
20. Elisabeth KMM, André M, Danny L et al. Comprehensive genetic diagnosis of acute myeloid leukemia by next-generation sequencing. Haematologica 2019;104:277-287.
21. Au CH, Wa A, Ho DN et al. Clinical evaluation of panel testing by next-generation sequencing (NGS) for gene mutations in myeloid neoplasms. Diagn Pathol 2016;11:11.
22. Blätte TJ, Schmalbrock LK, Skambraks S et al. getITD for FLT3-ITD-based MRD monitoring in AML. Leukemia 2019;33:2535-2539.
23. Wang T-Y, Yang R. ScanITD: Detecting internal tandem duplication with robust variant allele frequency estimation. GigaScience 2020;9.
24. Tsai HK, Brackett DG, Szeto D et al. Targeted Informatics for Optimal Detection, Characterization, and Quantification of FLT3 Internal Tandem Duplications Across Multiple Next-Generation Sequencing Platforms. J Mol Diagn 2020;22:1162-1178.
25. Kadri S, Zhen CJ, Wurst MN et al. Amplicon Indel Hunter Is a Novel Bioinformatics Tool to Detect Large Somatic Insertion/Deletion Mutations in Amplicon-Based Next-Generation Sequencing Data. J Mol Diagn 2015;17:635-643.
26. Levis MJ, Perl AE, Altman JK et al. A next-generation sequencing–based assay for minimal residual disease assessment in AML patients with FLT3-ITD mutations. Blood Adv 2018;2:825-831.






